I’m designing a Liquid Oxygen Bath to place composite coupons in the bath and soak 96 samples for intervals of 8 hours, 24 hours, 7 days, 21 days, 42 days, 62 days and 90 days. I’ve read the articles by Ralph Scurlock “Low Loss Dewars”, which has been very informative for the material selection of the bathtub. Due to the low emissivity of aluminum, I’ll use it for the bathtub.
The bathtub will be insulated with Perlite about 3″ thick and the outer bathtub will hold the 3″ Perlite insulation and then the bathtub will set in another aluminum container to use the vapor/boil-off to help chill/insulate the bathtub. Should I have a thicker insulation of Perlite? Is there a LOX compatibility problem with Perlite?
I also plan to introduce a very small purge of gaseous nitrogen to eliminate any moisture/ice build-up inside the bathtub to keep the stainless steel float valve from sticking. The bathtub will have an insulated lid on it, however, it will just lay on top there will be no confinement of vapor/boil-off.
Is there anything else I should take into consideration?



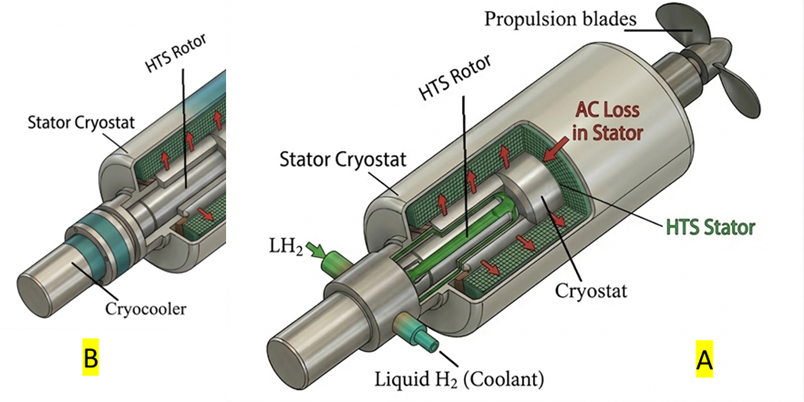





2 Comments
Dr. Peter Mason
August 1, 2008You don’t give an idea of the size, shape or desired hold time, so the following comments may be off base, but here goes.
1. I would consider a commercial dewar made by such companies as MVE, Cryofab, Cole Palmer, Janis or many other companies. If you can find the size you need, I would expect them to be far cheaper than a self-built unit. Many of these companies will fabricate to suit your needs also. Many of these can be bought with sample holders which may suit your purposes.
2. These dewars are typically made of stainless steel, which has far lower thermal conductivity than Al. This is more important than low emissivity, at least as far as hold time is concerned, because conduction around the walls is the major heat input.
3. Vacuum insulation will be far more efficient than perlite. Most commercial dewars are basically thermos bottles, double-walled, vacuum jacketed and sealed. The fancier ones use vapor-cooled shields to reduce hold time. They are about as efficient as you can get.
4. If you build the tub deep enough and close it with a reasonably efficient lid, the boil-off from the LOX bath will be an effective purge.
5. Make sure your design has a vent line to carry off the O2 to a safe place. It can be dangerous stuff if it concentrates in an area where there are flammables. Oil is particularly subject to flaming if there is a small spark or static discharge.
6. I would not use a float valve with a switch in the bath. Sounds like you might be planning en electric switch to control fill. Bad idea. See 5. If you do use a float, have it push a lever to the outside world. There are liquid level sensors which can be used to control a valve on your supply dewar to fill automatically.
Peter Mason
Physics Department
California Institute of Technology
626-395-3341
Dr. Randall F. Barron
August 1, 2008Perlite is a good cryogenic insulation and, because perlite is a silica-type material (glass-like), it is compatible with liquid oxygen. The only problems that have been reported in the literature were with opacified powders, in which small aluminum flakes were added to the powder—the aluminum oxidized (burned) very rapidly when oxygen was introduced, but the silica powder was not affected. [ R.F. Barron, CRYOGENIC SYSTEMS, 2nd ed., Oxford University Press, New York, (1985), pg. 396]
Many people have used a 4-inch (100mm) thick layer of closed-cell glass foam as an insulation for small containers used for cryogenic treatment of metals.
The glass foam is also compatible with LOX, and it has about the same thermal conductivity as gas-filled perlite (0.020 Btu/hr-ft-oF or 35 mW/m-k for glass foam and 0.015 to 0.025 Btu/hr-ft-oF or 26 to 44 mW/m-k for gas-filled perlite, depending on the density of the perlite). [R.F. Barron, CRYOGENIC HEAT TRANSFER, Taylor and Frances, Philadelphia, PA, (1999), pg. 25)
The 3-inch (76 mm) thick layer of perlite powder would be satisfactory, if vapor-cooling is used. In steady-state operation, you could expect about 8 to 10 L/hr (190 to 240 L/day) as the boil-off rate, if your container is on the order of 30”x 30”x 60” long (0.76 x 0.76 x 1.52 m long), with a volume of about 880 L. With a 4-inch (102mm) thick layer of perlite, the boil-off would be reduced to 6 to 8 L/hr.
Aluminum is a good material for cryogenic containers, particularly if it is important to maintain a uniform temperature over the entire wall of the container.
Aluminum does have the disadvantage that it will conduct heat very well from the warmer area near the top of the container to the liquid. You could achieve the same thermal radiation resistance by using 304 stainless steel for the container material and covering the surface with a layer or two of aluminized Mylar, since the emissivity is a surface effect—not a bulk effect. The stainless steel would conduct heat much less significantly from warm to cold regions.
There are some texts on design of small-scale cryogenic vessels that you might be interested in reading:
1. A. J. Croft, CRYOGENIC LABORATORY EQUIPMENT, Plenum Press, New York, (1970). See Chapter 2, Storage and Handling of Liquid Refrigerants; Chapter 7, Materials and Joining Methods; and Chapter 8, Cryostat Dewars.
2. J.G. Weisand, HANDBOOK OF CRYOGENIC ENGINEERING, Taylor and Frances, Philadelphia, PA, (1998). See Chapter 8, Cryostat Design.
3. G.K. White, EXPERIMENTAL TECHNIQUES IN LOW-TEMPERATURE PHYSICS, 3rd ed., Oxford University Press, New York, (1979). See Chapter VII, Introduction to Cryostat Design.
There’s also much helpful information in the volumes of ADVANCES IN CRYOGENIC ENGINEERING, vols. 1-48, Plenum Press, New York, (1965-2002).