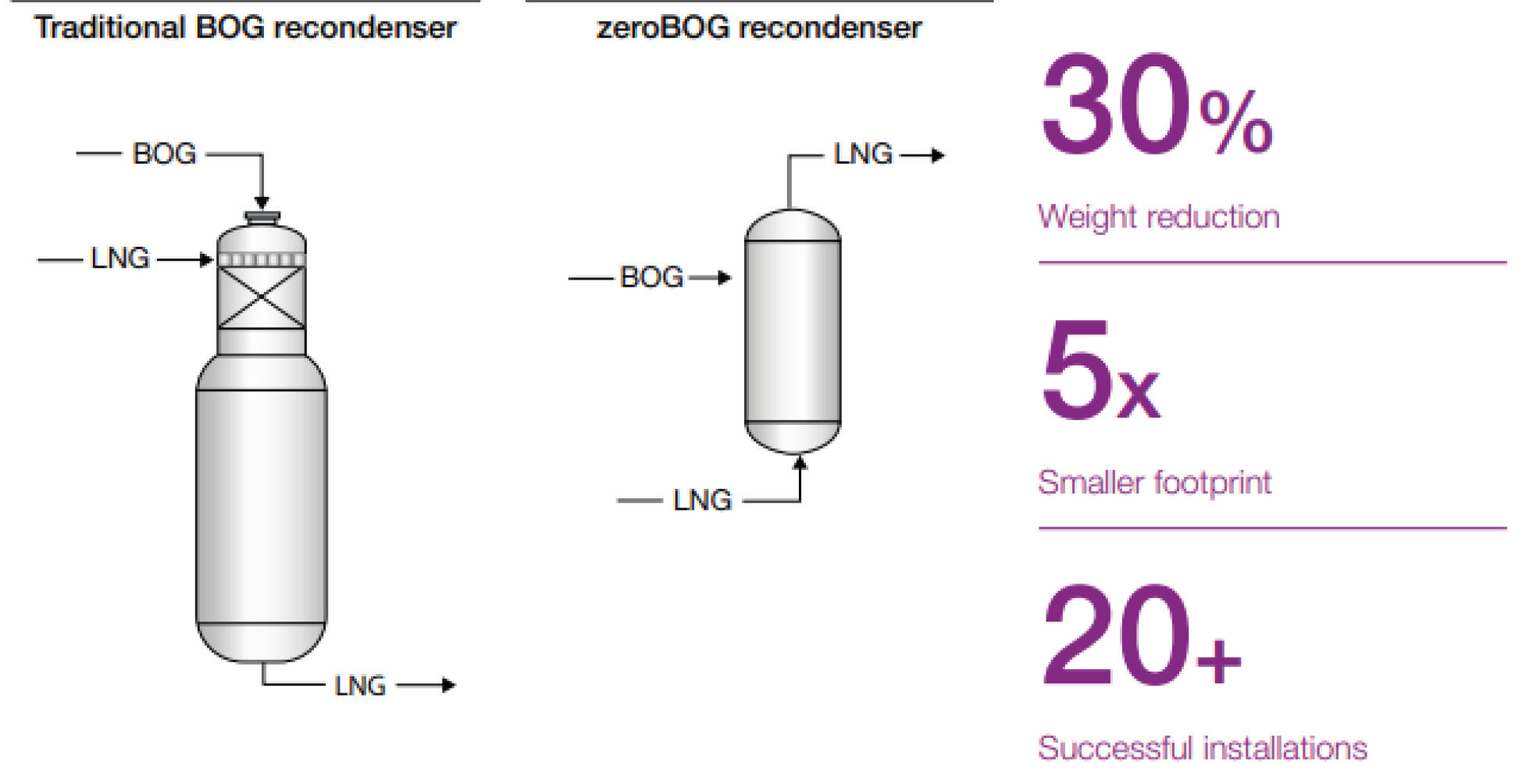
The Kelvin temperature scale is the principal temperature scale used in cryogenics and science. The kelvin is also one of the seven base units in the Système International d’Unités (SI) and is the unit of thermodynamic temperature. Thermodynamic temperature is a fundamental physical quantity (like mass, length or electrical current) and must equal zero at the point where entropy equals zero and effectively all motion stops. This point is known as absolute zero. It’s worth pointing out that the Third Law of Thermodynamics states that one can never reach absolute zero but can come vanishingly close.
All SI base units must be traceable back to a repeatable physical measurement or a single physical artifact. In order to understand the definition of the kelvin, it’s useful to look at its historical origins. In the nineteenth century, the British physicist William Thomson proposed a temperature scale in which a unit of heat transferred from an object at temperature T to an object at temperature T-1 would produce the same amount of work regardless of the value of T. Such a scale would be absolute (independent of any material properties). Thomson’s definition was actually part of a much larger debate over whether heat and work were in fact separate entities and was related to the development of the First and Second laws of Thermodynamics at that time. While changing the basis of the definition and the fixed point of his scale; Thomson kept the size of his temperature unit the same as that of a degree Celsius.
At this time, V. Regnault calculated for Thomson that absolute zero would correspond to -272.75°C [1]. Modern calculations fix this at -273.15°C. In recognition of his scientific and technical accomplishments; Thomson was ennobled as Baron Kelvin in 1892. His temperature scale is now known as the Kelvin scale.
Since we now know that we can’t reach absolute zero and thus can’t measure anything there, the fixed point for the Kelvin scale today is the triple point of pure water. This is the single temperature at which water can coexist as a solid, vapor and liquid. The SI unit kelvin is officially defined as: “The kelvin, unit of thermodynamic temperature, is the fraction 1/273.16 of the thermodynamic temperature of the triple point of water” [2]. In other words, the triple point of water occurs at 273.16K Since the triple point of water is also defined as 0.1°C, the conversion between Celsius and Kelvin is given by °C = K – 273.15.
A few comments on usage. Note that the unit is kelvin (lower case k). This usage is consistent with the American Institute of Physics style guidelines but is not universally applied; many publications still capitalize the unit. The symbol is K, not °K. Also, we should technically speak of 300 kelvins (just as we speak of 100 meters) but in conversation it’s almost always true that the singular form of the unit is used. Lastly, it’s really only convention that prevents us from discussing the weather using the Kelvin scale. As I write this, it is a pleasant 292K at my home in Pacifica CA.
More information about the Kelvin scale and SI units in general may be found in:
[1] Absolute Zero and the Conquest of Cold, T. Shachtman, Mariner Books (2000)