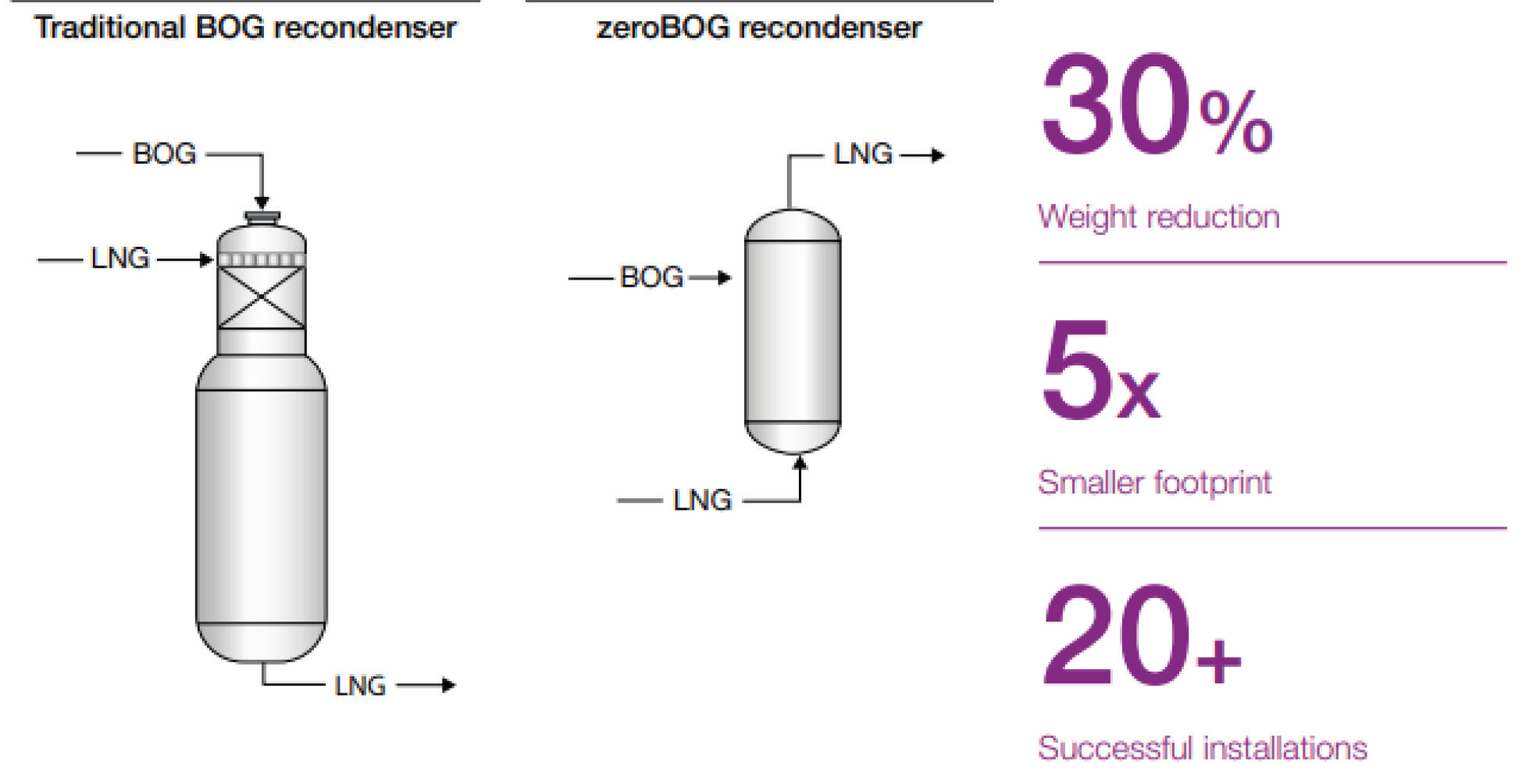
By Megan Burgasser
For the first time, researchers at the University of Cincinnati have visualized the structure of two key protein partners involved in inflammation and immune signaling. This breakthrough—published in the Proceedings of the National Academy of Sciences—not only uncovers the structure of the ADAM17 enzyme in complex with its regulatory partner iRhom2 but also marks a milestone for UC’s Center for Advanced Structural Biology (CASB), established just two years ago.
“This work provides a foundation for designing therapies targeting ADAM17-related diseases,” said Tom Seegar, Ph.D., an Ohio Eminent Scholar and assistant professor in the Department of Molecular and Cellular Biosciences at UC’s College of Medicine. “We’re offering new strategies to address critical health conditions.”
Seegar is the corresponding author on the study, the first published from his lab at UC. First authors include research scientist Joe Maciag, Ph.D. and graduate assistant Conner Slone. Their work provides a long-sought glimpse into how ADAM17 and iRhom2 interact—and how that interaction might be harnessed to better treat conditions such as rheumatoid arthritis, cancer and even Covid-19.
“If you can see something, you can figure out how it works,” said Seegar. “We are figuring out what this enzyme looks like and how it’s regulated.”
Why Structure Matters
ADAM17 is a membrane-bound enzyme found in every cell of the human body. It plays a vital role in shedding signaling molecules that control immune response and tissue repair. But when it malfunctions or becomes overactive—as it does in several chronic inflammatory diseases—the resulting immune signaling can become pathological.
Until now, researchers had only partial views of how this protein and its regulator behaved. “We know in some cancers and rheumatoid arthritis, way too much signaling is occurring,” said Maciag. “But some treatments create too many side effects, worse than the disease itself.”
To get a full picture, the Seegar Lab turned to a powerful new ally: cryogenic electron microscopy, or cryo-EM.
A New Microscope, A New Era
Launched in 2022, UC’s Center for Advanced Structural Biology was specifically designed to accelerate breakthroughs like this one. It houses two state-of-the-art electron microscopes and specialized instrumentation for sample preparation, vitrification and data processing.
“The Center for Advanced Structural Biology is a relatively new core facility that houses two state-of-the-art electron microscopes,” Seegar explained. “These high-magnification microscopes are optimized to visualize macromolecules embedded in thin layers of vitreous ice.”
Cryo-EM differs significantly from more traditional structural biology techniques such as X-ray crystallography, which requires large amounts of purified protein and the formation of crystals—a time-consuming and sometimes impossible feat for complex membrane proteins. Instead, cryo-EM uses rapid freezing to preserve biological samples in a close-to-native state.
“Cryo-EM typically requires less sample and avoids crystallization altogether,” Seegar noted. “The rapid freezing process preserves proteins in a near-native conformation, and when combined with single-particle analysis, can yield atomic-level resolution.”
That resolution—3.4 Ångströms[JS1] , or roughly the diameter of a water molecule—was critical to understanding how ADAM17 and iRhom2 communicate across the cell membrane and how disruptions in that communication may be implicated in disease.
Meeting the Challenge of Membrane Proteins
Integral membrane proteins like ADAM17 and iRhom2 are notoriously difficult to work with outside their native environments. That challenge was central to the UC team’s effort.
“We needed to develop purification strategies that maintained the proteins’ structural integrity outside of their native lipid context,” said Seegar. Once purified, the team optimized the freezing process and used a support grid to create a thin layer of vitreous ice suitable for high-resolution imaging.
To aid particle identification, they introduced an antibody fragment called a Fab, which helped orient the particles during data processing. “The cryogenic freezing process was critical,” Seegar said. “It preserved the proteins in their near-native conformations and prevented structural distortion or aggregation.”
The result: the first-ever detailed image of the ADAM17–iRhom2 complex and the identification of the iRhom2 “re-entry loop,” a structural element that transmits signals from the cell’s interior to its exterior and is required for ADAM17 to function.
On-Site Power: TEM + ARC
The team’s success also hinged on infrastructure beyond the microscope itself. UC’s 200 kV Thermo Fisher Glacios cryo-electron microscope and its 120 kV Talos L120C screening scope are supported by a full suite of sample preparation equipment and a staff trained in cryo-EM theory and technique. But what sets the center apart is its integration with UC’s Advanced Research Computing Cluster.
“This enables users to make immediate decisions on sample quality and collection strategy,” said Seegar. “Once data collection is complete, seamless transfer to the computing cluster allows for all downstream processing necessary to achieve atomic-resolution reconstructions.”
In short, everything—from freezing to processing to model reconstruction—was done on campus.
Building the Future
The Seegar Lab is now planning its next steps, including deeper studies into how adapter proteins like iRhom2 regulate enzymes such as ADAM17. “These adapter proteins are not well understood,” said graduate student Conner Slone. “Our research will be in understanding them and will be driven by specificity. Ideally, controlling these will allow researchers to control disease states.”
Other contributors to the study included Igal Ifergan, Ph.D., assistant professor in the Department of Molecular and Cellular Biosciences; graduate student Hala Alnajjar; Medical Scientist Training Program student Maria Rich; and undergraduate Bryce Guion. The study was funded by the National Institute of General Medical Sciences and supported by a University of Cincinnati Research Innovation/Pilot Grant.
Looking ahead, Seegar says the Center for Advanced Structural Biology will evolve alongside the needs of its users. “CASB is guided by an advisory committee that evaluates user needs and helps direct future instrumentation and workflow upgrades,” he said.
The cryo-EM center itself was made possible through funding from the NIH High-End Instrumentation Grant Program, the UC Office of the President and Vice President for Research and the College of Medicine.
“We are indebted to UC,” Seegar said. “Our work wouldn’t be possible without this research core facility.”